Select to expand quote
segler said..
There will not be any galvanic corrosion with either stainless or titanium screws.
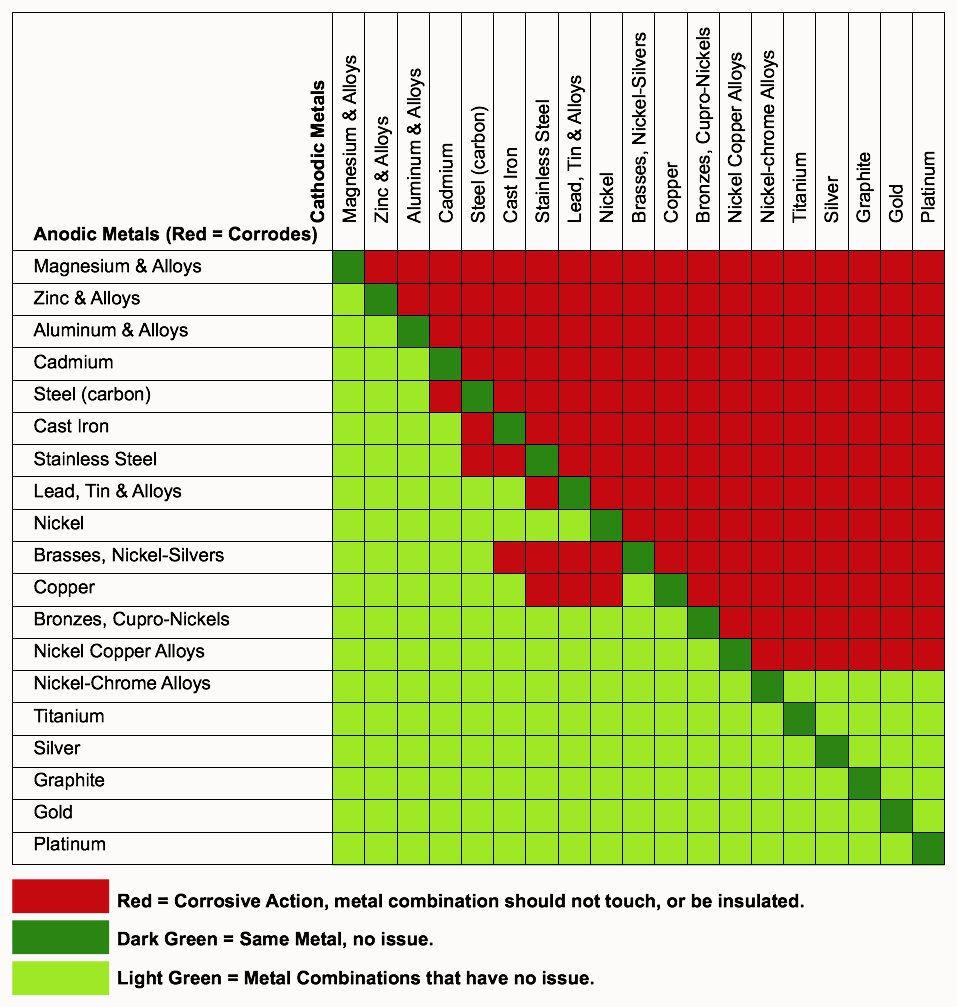
I had galvanic corrosion on my Streamlined mast extension and mast base, was between the respective anodized aluminum body and stainless steel screws. And titanium is certainly more stable than stainless steel, but is very reactive with oxygen, that is why titanium has to be welded in an oxygen free atmosphere. And the below colored chart indicates that aluminum and titanium do have a corrosion potential when aluminum is the anode.
For my Streamlined mast extension salt water could connect the underside of pan head SS screws and the anodized aluminum extension, because the extension body is round while the underside of the pan head screw is flat and so saltwater could bridge the two allowing for galvanic corrosion.
BEST METHODS TO PREVENT GALVANIC CORROSION between DISSIMILAR METALS:
Separate metals electrically by using non-conductive materials which stops ion migration:
Separate metals with an insulator like plastic; (this is what I did, used teflon tape and nylon washers)
Use water-repellent compounds like grease to prevent contact with electrolytes;
Consider electroplating using noble metals* (like gold, silver or platinum) that resist corrosion better.
Apply cathodic protection; This is the technique used to control the corrosion of a metal surface by making it the cathode of an electrochemical cell. A simple method of protection connects the metal to be protected (anode) to a more easily corroded 'sacrificial metal' to act as the anode. This is accomplished by physically connecting the anode that needs protecting by a wire to the sacrificial metal.
For Aluminum and Copper, apply antioxidant paste. Antioxidant paste can easily be found on the internet. It is sometimes referred to as 'connection grease.'