

Hey bro's
Here's a quick run down on corrosion basics for kite boarders.
I tried to upload it here but seabreeze doesnt seem to like lots of images so it crashed during the upload.
Here's the link to the topic on kiteforum. with images
kiteforum.com/viewtopic.php?f=1&t=2403406&p=1059384#p1059384
Hey bros,
Here's a quick run down on how corrosion works and how you can apply that knowledge to kiteboarding.
To transform metals from their natural state (eg iron ore)we expend large amounts of energy to form them into the metals that we know today. Once we have transformed the metals into the composition that we want they are known to be in a meta-stable state. The metal over time will shed energy and return to its original state. This is what we know as corrosion
All metals with the exception of gold have an oxide layer. That layer could be an active layer that continues to corrode. Eg iron oxide, rust.
Or a passive oxide layer could be formed that protects the underlying material. EG the chromium oxide layer found on stainless steels.
Corrosion is an electrochemical process.
Corrosion acts in the same way a battery works.
You need an anode, Cathode, an electrolyte solution, water and oxygen and temperature. If you can remove or reduce any of these factors you can reduce/slow the corrosion process!
kiteforum.com/download/file.php?id=88487&t=1
The difference between a battery and galvanic corrosion cell is that the anode and cathode are constantly touching each over. This allows the corrosion to propagate unchecked.
kiteforum.com/download/file.php?id=88490&t=1
All metals have voltage potential. When the voltage potential varies between the 2 metals we have a possibility of creating a galvanic cell.
The metal with the highest voltage becomes the cathode and the metal with the lowest voltage becomes the cathode. For galvanic corrosion to take place is standard services (eg in your house) you need a voltage potential difference of 0.5V.
However kitesurfing in the sea is not a standard service, It is considered a severe service. In a server service, the recommendation is to have no more than 0.15V potential difference between to two metals.
What is a server service? Service that increases the likelihood of corrosion. Increased temperature increases corrosion protential, increased moisture and increased reactivity in the elecrolyte solution eg salt water compared to fresh water
How do we know what voltage potential our metals have? Are they compatible? and will they be susceptible to corrosion.
We look at something called an anodic index or galvanic chart,
How do we know what voltage potential our metals have? Are they compatible? and will they be susceptible to corrosion.
We look at something called an anodic index or galvanic chart,
kiteforum.com/download/file.php?id=88491&t=1
If we take 2 common material used in kiteboarding. Aluminum and stainless. How far apart are these on the anodic index?
Much more than 0.15V.There will be galvanic corrosion in a server environment between aluminum and stainless steel.
Which material will corrode? the alloy or the stainless? The cathode will not corrode and the anode will. In this instance, the alloy is the anode and the stainless is the cathode.
How do we slow galvanic corrosion?
We can insulate the 2 dissimalar materials. If the 2 different metals are not touching then we have broken the battery connecttion much like turning off the switch. EG use a plastic washer.
Use materials within 0.15V potential differene.EG stainless with stainless.
Control the moisture between the 2 materials. Using a sealant or lubricant can keep moisture out off fasteners and prevent oxygen, moisture and elecrolyte solution.
Place a protective coating over the anode and cathode.
kiteforum.com/download/file.php?id=88492&t=1
So.... You say I don't have alloy to stainless, I only have stainless to stainless bolts. I wont get corrosion because we have the same voltage potential right?
Wrong You still have a chance for corrosion. This is where pitting and crevice corrosion come to play.
As mentioned above stainless steels have a protective oxide layer that prevents corrosion. This is because chromium has been added to the composition of the steel and creates the chromium oxide passive layer.
If the passive oxide layer breaks down and you have the reactive environment like sea water and warm temperatures then corrosion can take place. Interestingly Stainless steels have 2 voltage potentials. A voltage potential with a passive oxide layer and a lower voltage potential when the passive oxide layer breaks down. This means that a galvanic cell can be created on the surface of the stainless and a pit can start to be created. (38.5 KiB) Viewed 3 times
(38.5 KiB) Viewed 3 times
As the pit depth increases the corrosion products on the deprives the pit of oxygen removing the materials ability to regenerate its passive oxide layer. The pit becomes increasingly more anodic and reactive increasing the corrosion rate. Pitting corrosion is 10 x faster than general corrosion.
/images/misc/forum-image-missing.gif (27.26 KiB) Viewed 3 times
As you increase the temperature likelyhood of pitting corrosion increases, as you increase the reactivity of the electrolyte solution you increase the rate of pitting corrosion.
How do you prevent pitting corrosion?
Select appropriate materials that will work in the service you use.
Wash down after use in salt water, This removes the electrolyte solution
Keep gear stored dry,
Not all stainless steels are created equal. 304 and 316 are common grades used in kiteboarding.  (12.43 KiB) Viewed 3 times
(12.43 KiB) Viewed 3 times
As you can see in this critical pitting temperature chart,
304 SS has a low temperature of when pitting corrosion takes place.
316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible.
Lastly what is crevice corrosion? Crevice corrosion works in the same manner as pitting corrosion. However a the crevice deprives the material from oxygen and retains moisture and electrolyte solutions becoming increasingly more reactive far more quickly than pitting corrosion. Pitting corrosion is up to 100 times faster than general corrosion and starts at a lot lower temperature than pitting corrosion. (11.72 KiB) Viewed 3 times
(11.72 KiB) Viewed 3 times
How do we prevent pitting corrosion.
Also the same techniques as pitting corrosion.
We can also add drain holes so crevices can be flushed and washed out,
Sealant to prevent ingress into the crevice
On a design standpoint try to remove as many crevices from the design as possible.
So there you have it. A quick guide to corrosion. Does your brain hurt?
Here's a sumary, Corrosion works like a battery. If you can remove or reduce the factors that cause corrosion then you can slow the corrosion process.
Let me know if you have any questions.[
above the second last diagram u say wash down with salt water ??is this correct or do you mean fresh water
above the second last diagram u say wash down with salt water ??is this correct or do you mean fresh water
oops. wash down with fresh water.....
might need to explain this to foil manufacturers, but they should already know this.
titanium bolts into aluminium mast and fuselage. some paste and Teflon tape to protect it. They recommend dismantle after three uses in the ocean and reapply and wash with fresh water each use.
Yes, I know a full carbon fibre will get over this.
speaking my language Plummet, this is the field i work in
just one thing to add to the above
"316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible. "
316 and 316L are very good austenitic alloys for high corrosive areas BUT!! do not use a copper or zinc washer or antiseize with copper in it., it will fark your gear up, tefgel or similar is the best for this application
speaking my language Plummet, this is the field i work in
just one thing to add to the above
"316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible. "
316 and 316L are very good austenitic alloys for high corrosive areas BUT!! do not use a copper or zinc washer or antiseize with copper in it., it will fark your gear up, tefgel or similar is the best for this application
Hey Lost. What mechanism are you talking about re copper and stainless other than galvanic corrosion that should effect the copper and not the stainless?
I do recall something about copper/stainless embrittlement but thought this occurred at a lot higher temperatures than.
speaking my language Plummet, this is the field i work in
just one thing to add to the above
"316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible. "
316 and 316L are very good austenitic alloys for high corrosive areas BUT!! do not use a copper or zinc washer or antiseize with copper in it., it will fark your gear up, tefgel or similar is the best for this application
Hey Lost. What mechanism are you talking about re copper and stainless other than galvanic corrosion that should effect the copper and not the stainless?
I do recall something about copper/stainless embrittlement but thought this occurred at a lot higher temperatures than.
I believe the embrittlement issues for many austenitic grades begins at about 450 degC. hence why at high temps we use 321 and 253MA in furnaces.
I think the avoiding copper and zinc is a galvanic corrosion thing where the SS becomes the cathode thus generating hydrogen gas then the potential of hydrogen embrittlement becomes a factor? but I'm not a metallurgist. (my dad is however)
Way over my head i just make a little loop do a turn and a half nip it off and shuffle down a couple squares
Im looking forward to plumbing with metalmet once the gin wears off tomorrow 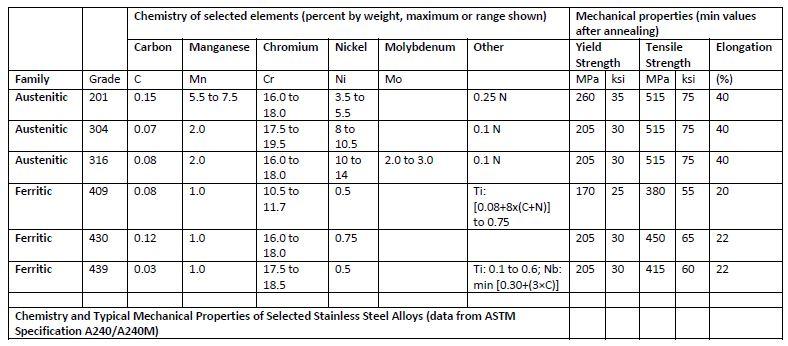
wait let me get this right:
Hey bros, kite gear not like salt water, salt water bad. Wash gear with tap - gear last more longa, when gear break buy new gear.
wait let me get this right:
Hey bros, kite gear not like salt water, salt water bad. Wash gear with tap - gear last more longa, when gear break buy new gear.
Don't forget inspect gear, dry gear, store gear, lubricate gear, clean gear, seal gear, buy similar voltage potential gear.
wait let me get this right:
Hey bros, kite gear not like salt water, salt water bad. Wash gear with tap - gear last more longa, when gear break buy new gear.
Yeah we get it. hes from new zealand. We let him get away with this stuff tho
Just curious, do I need a conversion table to account for the higher salt content of the South European Atlantic coast ?
Just curious, do I need a conversion table to account for the higher salt content of the South European Atlantic coast ?
Increasing the chloride content will increase the the corrosion rate and decrease the temperature at which pitting and crevice corrosion take place.
In short, wash,dry, seal, replace **** more often.
Am liking this subject, thanks - on top of salty seas, the fresh water for cleaning here has very high sulphur content too (deep ground water wells) ... being stuck between 2 evils, tend to just let the kit dry in the warm air .... seems to work but curious for any thoughts on the chemical intelligence of that ? main concern has been for the kit stitching.
Am liking this subject, thanks - on top of salty seas, the fresh water for cleaning here has very high sulphur content too (deep ground water wells) ... being stuck between 2 evils, tend to just let the kit dry in the warm air .... seems to work but curious for any thoughts on the chemical intelligence of that ? main concern has been for the kit stitching.
Sulphur can lead to sulphide stress cracking. But I really doubt that be an issue.
Sorry, I can't help te stitching.
speaking my language Plummet, this is the field i work in
just one thing to add to the above
"316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible. "
316 and 316L are very good austenitic alloys for high corrosive areas BUT!! do not use a copper or zinc washer or antiseize with copper in it., it will fark your gear up, tefgel or similar is the best for this application
Hey Lost. What mechanism are you talking about re copper and stainless other than galvanic corrosion that should effect the copper and not the stainless?
I do recall something about copper/stainless embrittlement but thought this occurred at a lot higher temperatures than.
Metallurgy is a fairly complex beast at times
To break it down...it will occur at all temps but the chemical reaction is different in different temp ranges. The galvanic corrosion (form of electrolysis) is accelerated by electrolytes (salt water) and attacks the stainless more than the copper. The root of the tread is a perfect stress riser for stress corrosion cracking. There's some interesting stuff that also happens from the metal metal contact from screwing in a bolt, it can generate bull****e high temps from the friction changing the hardness/tensile strength of bolts, making it more likely to crack. If your interested I'll chase up some old metallurgical macro reports I done on old windsurfing fin bolts
If you get really bored plummet Google microbial influenced corrosion
speaking my language Plummet, this is the field i work in
just one thing to add to the above
"316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible. "
316 and 316L are very good austenitic alloys for high corrosive areas BUT!! do not use a copper or zinc washer or antiseize with copper in it., it will fark your gear up, tefgel or similar is the best for this application
Hey Lost. What mechanism are you talking about re copper and stainless other than galvanic corrosion that should effect the copper and not the stainless?
I do recall something about copper/stainless embrittlement but thought this occurred at a lot higher temperatures than.
Metallurgy is a fairly complex beast at times
To break it down...it will occur at all temps but the chemical reaction is different in different temp ranges. The galvanic corrosion (form of electrolysis) is accelerated by electrolytes (salt water) and attacks the stainless more than the copper. The root of the tread is a perfect stress riser for stress corrosion cracking. There's some interesting stuff that also happens from the metal metal contact from screwing in a bolt, it can generate bull****e high temps from the friction changing the hardness/tensile strength of bolts, making it more likely to crack. If your interested I'll chase up some old metallurgical macro reports I done on old windsurfing fin bolts
If you get really bored plummet Google microbial influenced corrosion
Yeah. I get where you are going with this. A perfect storm of all sorts of corrosion going on in the fastener crevice.... I understand SCC and MIC. I omitted SCC and MIC and IGC from the write up... Cauncy would litterally throw his toys out of the cot with. But maybe I should upload some basic info on those too.
But do chase up your old reports on windsurfing bolts. I am interested in reading those.
speaking my language Plummet, this is the field i work in
just one thing to add to the above
"316 includes 2.0-2.5% molybdenum. This increases corrosion resistance. When selecting stainless parts for kiteboarding. Try to select 316 parts where possible. "
316 and 316L are very good austenitic alloys for high corrosive areas BUT!! do not use a copper or zinc washer or antiseize with copper in it., it will fark your gear up, tefgel or similar is the best for this application
Hey Lost. What mechanism are you talking about re copper and stainless other than galvanic corrosion that should effect the copper and not the stainless?
I do recall something about copper/stainless embrittlement but thought this occurred at a lot higher temperatures than.
Metallurgy is a fairly complex beast at times
To break it down...it will occur at all temps but the chemical reaction is different in different temp ranges. The galvanic corrosion (form of electrolysis) is accelerated by electrolytes (salt water) and attacks the stainless more than the copper. The root of the tread is a perfect stress riser for stress corrosion cracking. There's some interesting stuff that also happens from the metal metal contact from screwing in a bolt, it can generate bull****e high temps from the friction changing the hardness/tensile strength of bolts, making it more likely to crack. If your interested I'll chase up some old metallurgical macro reports I done on old windsurfing fin bolts
If you get really bored plummet Google microbial influenced corrosion
Yeah. I get where you are going with this. A perfect storm of all sorts of corrosion going on in the fastener crevice.... I understand SCC and MIC. I omitted SCC and MIC and IGC from the write up... Cauncy would litterally throw his toys out of the cot with. But maybe I should upload some basic info on those too.
But do chase up your old reports on windsurfing bolts. I am interested in reading those.
Great topic and info!
Could you also include the reasons for "Galling" and how to avoid it, we see a fair number of snapped off screws in the inserts of TT's (we assemble boards with Tefgel and have never had an issue since switching to this stuff)?
Ok, what is Intergranular corrosion?
Intergranular corrosion is the preferential attack of the grain boundaries within the metal crystal structure. Why does it happen and how can we guard against it?
When stainless steels are heated for between 500 and 800 deg C for a period of time they can become sensitized. Carbon can start to precipitate and joins with chromium to cause chromium carbide precipitation. The net effect once the steel has cooled down is chromium depleted zones in the grain boundaries. This means at those zones the material has lower corrosion resistance. In the right enviroment corrosion can start and propogate through the grain boundries of the steel.

This typically takes place when welding in the heat affected zones around the weld if the weld has been keep hot for too long.
How do we prevent intergranular corrosion?
By far the easiest method is to control the chemistry of the steel you are welding.
Standard 316 will take approximately one minute of heating into the critical zone to be susceptible to chromium carbide precipitation.
However if you specify 316L low carbon content stainless then it will take 10 minutes approximately to be susceptible chromium carbide precipitation. Since most welded stainless widgets are small and will not be welded for more than 1 or 2 mins selecting 316L is the easiest method to guard against intergranular corrosion.
However, as a consumer you do not control the material selection process.
What else can you do? Heat treatment of parts to redissolve the chromium is an option. But this is not cost effective for a one-off part,
You can follow your washdown/dry proceedure for these parts and do periodic inspection and replacement. If a part shows corrosion on or by a weld replace the part asap.

What is MIC?
Mic stands for microbial induced corrosion.
Essentially when microbes attached together to form a bacterial colony on the metal surface they can create the conditions that lead to pitting corrosion.
You end up with a localized anodic zone created by the bacteria. Chemical reactions of the bacterial can also accelerate the corrosion process.

If you want more detail watch this vid
What is SCC?
SCC stands for Stress Corrosion Cracking.
When you combine a susceptible material, corrosive environment and stress/strain you have the conditions for SCC.
Stainless steels are a susceptible material, Saltwater is a corrosive environment that could enable SCC. Typically SCC occurs over 55 Deg C. However it is possible at lower temps if the environment and stress are sufficient.
Now you add tensile stress. This could be stress or stain caused by the parts use or manufacturing stresses like welding, forming etc.
SCC typically starts with stress/strain breaking the passive oxide layer at the crack tip exposing active material to the corrosive environment and corrosion starts. If the stress is above a threshold level then the crack will continue to propagate exposing more active material leading to more corrosion.
Interestingly the SCC occurs well below the yield strength of the material. So parts can fail well below the designers safety margins. What is yield strength you ask? Yeild strength where a material goes from elastic deformation to plastic deformation. IE bend it and it bounces back = elastic deformation, bend it and it stays bent = plastic deformation.

Perhaps my failed stainless widget has failed due to stress corrosion cracking? or a combination of SCC and IGC?

What are the corrosion controls for MIC
Clean and lubricate your gear, make sure there is no build up of foreign material that could lead to a colony of bacteria.
SCC?
Wash down your gear and don't leave salty damp gear in a hot car for hours on end. Remember that corrosion takes place faster when the temperature increases. For SCC it may not occur at ocean temps. However if you then stuff your gear into the car and leave it there in the heat of the summer you could take the parts into the danger zone for scc.
This is like being back at school
FuC#ing Shidt
I'm with you mick and I did a metallurgy degree looking closely at processes such as electrolysis... bad flash backs pouring over complex phase diagrams ![]() ... please stop. Lube your ass then wash your Sh1t... done.
... please stop. Lube your ass then wash your Sh1t... done. ![]()